Prolactin and Growth Hormone Pathophysiology
Our team specializes in translational approaches to unravel the intricate cellular and molecular mechanisms driving the progression and treatment resistance of breast and prostate cancers, with a particular focus on cytokine/hormone signaling and metabolic (iron) regulation of epithelial cell stemness and plasticity.
As we face the prospect of increasing cancer incidence due to extended lifespans, we emphasize the importance of early diagnosis and intervention. For localized breast and prostate tumors, surgical and radiotherapeutic treatments, combined with proactive preventive measures like PSA screening and mammography, have proven effective. However, as these diseases advance to more complex stages, the standard of care transitions to medical treatments such as anti-androgen and anti-estrogen therapy and chemotherapy. Unfortunately, the emergence of treatment resistance is a common challenge, often leading to the progression of these cancers to metastatic stages. Drug-induced tumor cell plasticity is a key mechanism of tumor relapse.
Our research primarily revolves around three central themes:
1- Understanding Prostate Cancer Progression (Vincent Goffin, Jacques-Emmanuel Guidotti and Charles Dariane)
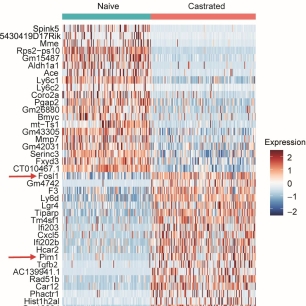
Our goal is to investigate and target the mechanisms by which castration-tolerant prostatic progenitor-like cells contribute to therapeutic resistance in prostate cancer. This research program gathers biologists, urologists, uro-pathologists and bioinformaticians, and involves various preclinical models including transgenic mice and patient -derived prostate tumoroids. It is currently supported by grants from the National Cancer Institute (INCa), University Paris Cité (Emergence Idex 2025), and Bayer company.
2- Understanding Benign Prostate Hyperplasia (Vincent Goffin, Nicolas Barry-Delongchamps and Julien Anract)
Our goal is to map the molecular and cellular interactions between prostatic progenitor-like cells and immune cells of the micro-environment in benign prostate hyperplasia, with particular focus on STAT5 signaling. This research program gathers biologists, immunologists, urologists, uro-pathologists and bioinformaticians, and involves various preclinical models including a unique BPH mouse model and patient-derived benign prostate organoids. It is currently supported by a grant from the National Research Agency (ANR).
3- Advancing Breast Cancer Research (Ahmed Hamaï, FEROSTEM group)
In this project, we are delving into iron metabolism and the molecular mechanisms triggering ferroptosis (constituting a therapeutic vulnerability) in breast cancer stem cells (BCSCs). Additionally, we will explore the role of autophagy in stemness-associated plasticity using preclinical models of breast cancer. To go further, our aim is to dissect the role of iron metabolism and autophagy in the crosstalk between BCSCs and the stromal/tumor immune microenvironment. By combining integrated bioinformatics with omics approaches, we want to identify innovative prognostic signatures and therapeutic targets at the molecular/cellular scale. In collaboration with clinicians, our work could lead to new effective therapies by targeting CSCs for breast cancer patients and for long-term tumor inhibition. This project has been supported for several years by the 'Ligue contre le cancer', and University Paris Cité (Emergence Idex 2025).
4- Understanding Prolactin Actions in Adrenarche (Vincent Goffin)
In line with our longstanding interest in understanding the pleiotropy of prolactin, the aim of this new collaborative project is to elucidate the role of prolactin in the process of physiological and premature adrenarche (androgen production by the adrenal cortex). Since children with premature adrenarche are at increased risk of cardiometabolic disorders in adulthood, one of the main objectives is to identify biomarkers of prolactin action in the adrenal gland in order to improve early diagnosis. This ANR-supported project involves basic and clinical endocrinologists.